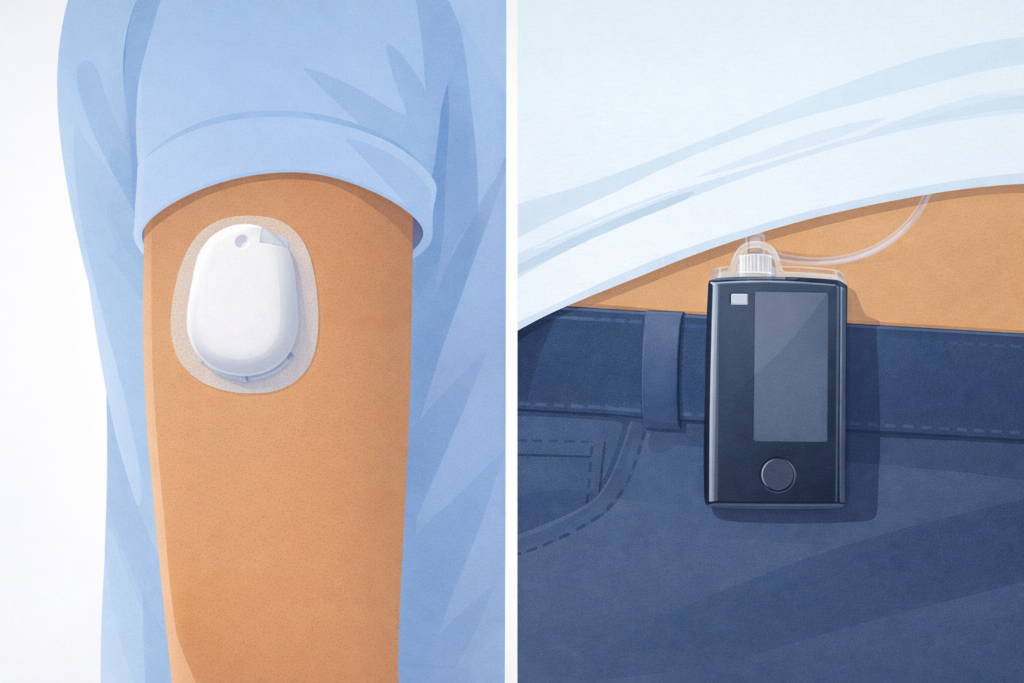
The best insulin pumps of 2026 offer more automation, more CGM options, and more lifestyle flexibility than ever before — and for the first time, automated insulin delivery (AID) is now the recommended standard of care for people with type 1 diabetes and for those with type 2 diabetes on multiple daily injections (ADA Standards of Care 2026: Section 7). Whether you’re newly diagnosed or a long-time pump user, this guide compares every major insulin pump available in the U.S. today — specs, CGM compatibility, AID features, and who each system is best suited for.
The best insulin pumps of 2026 offer more automation, more CGM options, and more lifestyle flexibility than ever before — and for the first time, automated insulin delivery (AID) is now the recommended standard of care for people with type 1 diabetes and for those with type 2 diabetes on multiple daily injections (ADA Standards of Care 2026: Section 7). Whether you’re newly diagnosed or a long-time pump user, this guide compares every major insulin pump available in the U.S. today — specs, CGM compatibility, AID features, and who each system is best suited for.
The good news: there are more options than ever, and newer systems are truly easier to use. Here’s what you should know before making your next choice.
→ Jump to Frequently Asked Questions
What Are the Top Insulin Pumps of 2026?
The tables below list all currently available insulin pumps in the U.S., organized by whether they include automated insulin delivery. All blood sugar values are in mg/dL, with mmol/L in parentheses where noted.
Automated Insulin Delivery (AID) Systems — Currently Available in the U.S.
| Pump | AID Algorithm | CGM Compatible | Best For |
|---|---|---|---|
| Tandem t:slim X2 | Control-IQ+ | Dexcom G6/G7, FreeStyle Libre 3 Plus | Adults & teens wanting touchscreen + AID |
| Tandem Mobi | Control-IQ+ | Dexcom G6, Dexcom G7 (10-day) | Adults wanting the smallest durable AID pump |
| Omnipod 5 | Omnipod SmartAdjust | Dexcom G6/G7, Libre 2 Plus; Libre 3 Plus rollout expanding (verify current availability) | Tubeless AID for T1D (age 2+) and T2D adults |
| Medtronic 780G | SmartGuard | Guardian 4, Simplera Sync, Instinct (Abbott) | Users who prefer the Medtronic/MiniMed ecosystem |
| iLet Bionic Pancreas | iLet algorithm (no carb counting) | Dexcom G6/G7, Libre 3 Plus (supported configs) | Users who want minimal meal management |
| twiist (Sequel Med Tech) | Tidepool Loop (Diabeloop DBLG2 option in 2026) | FreeStyle Libre 3 Plus, Eversense 365 | Tech-forward users; iPhone + Apple Watch control |
Non-AID Systems & DIY Options
| Pump | Type | CGM Integration | Best For |
|---|---|---|---|
| Omnipod Dash | Tubeless, manual | None (manual bolus only) | Tubeless delivery without CGM requirement |
| Dana Diabecare IIS* | Tubed, manual/DIY | Via third-party DIY apps only | DIY Loop users; primarily international markets |
| Loop (DIY) | Open-source AID | Multiple (Dexcom, Libre, others) | Tech-savvy users willing to build their own system |
*Dana Diabecare IIS has limited commercial availability in the U.S. and is not a mainstream option for most U.S. patients. See the DIY/International Options section below.
How Do Insulin Pumps Compare on Key Features?
Use the tables below to compare the technical details that matter most.
Size, Weight & Water Resistance
| Pump | Size | Weight | Water Rating |
|---|---|---|---|
| Tandem Mobi | 2.02″ × 1.47″ × 0.56″ | 1.06 oz (full) | IP28 (8 ft / 2 hrs) |
| Tandem t:slim X2 | 3.13″ × 2.0″ × 0.6″ | 3.95 oz (full) | IP27 (3 ft / 30 min) |
| Omnipod 5 / Dash Pod | 1.53″ × 2.05″ × 0.57″ | 0.88 oz (25 g) | IP28 (25 ft / 60 min) |
| Medtronic 780G | 2.1″ × 3.78″ × 0.96″ | 5.6 oz (158.8 g) | IP22 (drip-resistant only) |
| iLet Bionic Pancreas | Smartphone-sized | ~5 oz | IPX7 (3 ft / 30 min) |
| twiist | ~Oreo cookie size | < 2 oz | IPX8 (see manufacturer) |
| Dana Diabecare IIS | 2.17″ × 3.78″ × 0.87″ | 2.8 oz (80 g) | IPX8 (12 ft / 60 min) |
| Loop (DIY) | Varies by hardware | Varies | Varies by hardware |
Reservoir, Basal & Bolus Delivery
| Pump | Reservoir | Basal Range (units/hr) | Min. Bolus |
|---|---|---|---|
| Tandem Mobi | 200 units | 0.1 – 15 | 0.05 units |
| Tandem t:slim X2 | 300 units | 0.1 – 15 | 0.05 units |
| Omnipod 5 / Dash | 200 units | 0.05 – 30 | 0.05 units |
| Medtronic 780G | 300 units | 0.025 – 35 | 0.025 units |
| iLet Bionic Pancreas | 200 units | 0.05 – 30 | 0.05 units |
| twiist | 300 units (disposable cassette) | Configurable via algorithm | < 1 unit (microdose) |
| Dana Diabecare IIS | 300 units | 0.04 – 25 | 0.04 units |
| Loop (DIY) | Depends on pump hardware | Configurable | Configurable |
How Does Each Insulin Pump Work?
1. Tandem Mobi: The Smallest Durable AID Pump
The Tandem Mobi is the smallest durable (reusable) AID pump available. Slightly over 2 inches long and weighing only 1.06 oz, it easily fits in a pocket or can be worn directly on the body with the Tandem adhesive sleeve, using just 5 inches of tubing. It pairs with the Dexcom G6 or standard Dexcom G7 (10-day) continuous glucose monitor (CGM) and uses the Control-IQ+ algorithm to automatically adjust your basal rate and deliver correction boluses based on your blood sugar trends.
Who it’s best for: People who want the smallest durable AID pump, prefer smartphone control, and are comfortable with a tubed system.
Limitations: Needs a compatible iPhone or Android device for full control. Not tubeless. CGM compatibility is limited to Dexcom G6 and standard Dexcom G7 — does not currently work with FreeStyle Libre sensors.
2025–2026 Updates: The Android app received FDA approval and will launch in 2026. Tandem reported FDA clearance in late 2025 for extended-wear use of the SteadiSet infusion set, with a wider commercial rollout expected afterward — potentially halving site change frequency.
2. Tandem t:slim X2: Touchscreen AID with Expanded CGM Options
The t:slim X2 features a bright touchscreen display and uses the same Control-IQ+ algorithm as the Mobi. Its larger 300-unit reservoir is ideal for individuals with higher insulin needs. The t:slim X2 is compatible with Dexcom G6, Dexcom G7, and FreeStyle Libre 3 Plus in the U.S., offering users the widest CGM options among any Tandem pump.
Who it’s best for: People who prefer a touchscreen interface, want a 300-unit reservoir, and seek flexibility between Dexcom and FreeStyle Libre CGMs.
Limitations: Larger than the Mobi; tubed design; requires regular USB-C charging.
3. Omnipod 5: The Leading Tubeless AID System
The Omnipod 5 is the most popular tubeless AID system in the U.S. Its waterproof pod (IP28, rated for 25 feet for 60 minutes) attaches directly to your skin without tubing. The SmartAdjust algorithm automatically modifies insulin delivery based on real-time CGM readings. Omnipod 5 is also FDA-approved for adults with type 2 diabetes who use insulin.
Omnipod 5 currently supports Dexcom G6, Dexcom G7, and FreeStyle Libre 2 Plus. Newer company materials suggest a FreeStyle Libre 3 Plus rollout with limited or expanding availability. Check current Omnipod compatibility resources and confirm with your provider or pharmacy before prescribing.
Smartphone vs. controller: Omnipod 5 can be controlled with the compatible smartphone app or the Omnipod PDM controller — a smartphone is not necessary.
Who it’s best for: Active users, swimmers, children (FDA-cleared from age 2), and anyone seeking tubeless freedom with full AID automation.
Limitations: Pod needs replacement every 3 days. CGM required for SmartAdjust automation.
Near-term 2026 update: Insulet’s key upcoming U.S. development is an Omnipod 5 algorithm upgrade that will lower the target to 100 mg/dL (5.6 mmol/L) in 2026.
4. Omnipod Dash: Tubeless and No CGM Needed
The Omnipod Dash uses the same tubeless pod as the Omnipod 5, but it lacks CGM integration and automated insulin delivery. You manually manage all boluses with the PDM or the Omnipod app. This can be a good starting point for users who want tubeless delivery without committing to a full AID system.
Who it’s best for: People who want tubeless insulin delivery without CGM integration or who are not yet ready for AID.
Limitations: No automated adjustments; no CGM integration; all boluses are manual.
5. Medtronic 780G (MiniMed): Refined Automation
The Medtronic 780G, now branded MiniMed, provides some of the most precise basal adjustments of any pump — as low as 0.025 units per hour — making it an excellent choice for individuals with low or highly variable insulin needs. The SmartGuard algorithm can automatically deliver correction boluses, in addition to adjusting basal rates. In September 2025, Medtronic received FDA clearance to expand the 780G to include adults with type 2 diabetes on insulin.
CGM compatibility for the 780G is limited to Medtronic’s own ecosystem: the Guardian 4, Simplera Sync, and the newer Instinct sensor (similar to Abbott’s FreeStyle Libre 3+). The 780G does not work with Dexcom or FreeStyle Libre sensors.
Who it’s best for: Users who want very fine insulin-increment control and are comfortable with the Medtronic/MiniMed ecosystem.
Limitations: IP22 water resistance — not suitable for swimming. Larger and heavier than most options. Requires Medtronic-compatible CGM sensors only.
6. iLet Bionic Pancreas: Simple Setup, Fully Automated
The iLet adopts a different approach to AID: instead of counting carbohydrates in grams, you simply indicate whether your meal is small, medium, or large. The algorithm handles the rest, adapting over time to your insulin needs. As of 2025, the iLet is compatible with Dexcom G6, Dexcom G7, and FreeStyle Libre 3 Plus in supported configurations. This makes it a strong option for anyone exploring insulin therapy who wants fewer daily decisions.
Who it’s best for: People who struggle with carb counting or want the most hands-off AID experience available.
Limitations: Less detailed control for users who want precise carb entry. A dual-hormone (insulin + glucagon) version is in development but not yet widely available.
7. twiist (Sequel Med Tech): Newest AID System with iPhone and Apple Watch Control
The twiist is the newest FDA-cleared AID system on the U.S. market, launched July 7, 2025. Developed by Sequel Med Tech using DEKA’s pump technology, twiist employs the Tidepool Loop algorithm — originally created by the diabetes community — along with a new sound-wave technology called iiSure to directly measure every microdose of insulin delivered. This enables it to detect blockages (occlusions) up to 9 times faster than other pumps.
Roughly the size of an Oreo cookie and weighing less than 2 oz, twiist holds 300 units of insulin in a disposable cassette attached to a reusable pump body designed to last up to 3 years. The system is primarily controlled via iPhone, with the option to also manage it from a connected Apple Watch — making it the first FDA-cleared AID system with Apple Watch control capability. Users can set blood sugar targets as low as 87 mg/dL (4.8 mmol/L), the lowest of any commercial AID system. A meal presets feature allows you to choose fast, standard, or slow meal absorption using food emojis. Users can also retroactively adjust a carb entry after a meal if plans change mid-eating.
Who it’s best for: Tech-savvy users seeking the latest algorithm, iPhone control with optional Apple Watch access, and the most accurate dosing. Also ideal for those who want pharmacy access without a long-term commitment.
Limitations: Currently requires an iPhone (Android version in development). Cassette needs replacement every 3 days. FDA-cleared for use with type 1 diabetes only, for ages 6 and up.
CGM Compatibility: FreeStyle Libre 3 Plus and Eversense 365 (the 1-year implantable sensor).
Cost: Limited to $50/month with eligible insurance. The first month of supplies, including the pump, is free. Available at pharmacies.
📝 2026 Update: Sequel announced an optional algorithm — Diabeloop’s DBLG2 — which requires only your weight and total daily insulin. No carb entry needed.
8. Dana Diabecare IIS and Loop (DIY): Options for Experienced Users
The Dana Diabecare IIS is a compact tubed pump mainly used by individuals creating their own closed-loop systems with open-source apps. It has limited commercial distribution in the U.S. and is not a common choice for most U.S. patients — it is mostly used in international markets and by the DIY diabetes community. If you are considering DIY systems, the Dana IIS provides good water resistance (IPX8) and a long battery life (up to 14 days), but it requires substantial technical setup.
Loop is a free, open-source AID system developed by the diabetes community. It connects a compatible pump and CGM using an algorithm running on an iPhone, while AndroidAPS serves a similar purpose for Android users. In 2026, both DIY Loop and AndroidAPS have been updated. The ADA 2026 Standards of Care note that when patients use open-source AID, clinicians should support them safely and non-judgmentally. Loop is not FDA-approved as a complete system and requires ongoing technical maintenance — it is not recommended as a first choice for people seeking a plug-and-play experience.
The following products have been announced but are not yet commercially available in the U.S. This section will be updated as launch dates are confirmed.
- MiniMed Flex (Medtronic): A smaller, screenless version of the 780G, controlled via iPhone or Android. Scheduled for launch by summer 2026. Uses the same SmartGuard algorithm and 300-unit cartridge as the 780G. Current 780G buyers between Feb. 18, 2026, and the Flex launch may qualify for a $0 upgrade through Medtronic’s Forward Program.
- Omnipod 6 (Insulet): A future platform featuring an adaptive learning algorithm and compatibility with multiple CGMs. Investor materials indicate a 2027 timeframe.
- Tandem Tubeless Mobi: A tubeless version of the Mobi pump, expected to feature the Control-IQ+ algorithm and attach to a body-worn patch. Timeline not yet confirmed.
- Beta Bionics Mint: A tubeless, phone-free patch pump requiring no charging, using disposable batteries. 3-day wear time, 200-unit capacity. Expected in 2027.
- Sigi (Tandem): A tubeless-first pump with a reusable body and swappable insulin cartridges. Under development.
Tubed vs. Tubeless: Which Insulin Pump Style Is Right for You?
| Factor | Tubed Pump | Tubeless Pod |
|---|---|---|
| Reservoir size | Up to 300 units (Tandem X2, 780G, twiist) | Up to 200 units (Omnipod) |
| Freedom of movement | Tubing can snag or pull | No external tubing; fully wearable |
| Water resistance | Tandem Mobi IP28; 780G IP22 only | Omnipod IP28 (best-in-class) |
| Site changes | Every 2–3 days; extended sets clearing for longer wear | Every 3 days |
| Discreetness | Pump worn in pocket or clipped on | Pod sits flat on skin under clothing |
| CGM flexibility | t:slim X2 supports Dexcom and Libre sensors | Omnipod 5 supports Dexcom and Libre 2 Plus (Libre 3 Plus expanding) |
Which CGMs Are Compatible with Each Insulin Pump?
CGM compatibility determines whether automated insulin delivery works. Here’s where things stand in 2026. Always confirm current compatibility with the manufacturer before prescribing, since rollouts can be phased. For a deeper look at sensor options, see our guide to continuous glucose monitors.
| CGM Sensor | Compatible Pumps |
|---|---|
| Dexcom G6 | Tandem t:slim X2, Tandem Mobi, Omnipod 5, iLet |
| Dexcom G7 (standard 10-day) | Tandem t:slim X2, Tandem Mobi, Omnipod 5 (app), iLet |
| Abbott FreeStyle Libre 2 Plus | Omnipod 5 (controller), Tandem t:slim X2 |
| Abbott FreeStyle Libre 3 Plus | Tandem t:slim X2, iLet, twiist; Omnipod 5 (rollout expanding — verify) |
| Medtronic Guardian 4 / Simplera Sync / Instinct | Medtronic 780G only |
| Eversense 365 (1-year implant) | twiist (2026) |
What Does the ADA Recommend for Insulin Pumps in 2026?
The American Diabetes Association’s Standards of Care 2026 (Section 7: Diabetes Technology) includes several important updates that affect how and when pumps are prescribed.
- AID is now the preferred method for insulin delivery among people with type 1 diabetes and for those with type 2 diabetes on multiple daily injections who are not achieving their blood sugar goals.
- No gatekeeping: There is no minimum C-peptide level, auto-antibody test, or specific time on insulin required before starting an AID system. AID can be offered at or shortly after diagnosis.
- CGM at diagnosis is recommended, not postponed. Early access to CGM and AID is linked to improved long-term outcomes.
- Open-source AID: When patients use open-source AID systems, clinicians should support them safely and non-judgmentally — the ADA recognizes their use and encourages proper clinical oversight.
- Basal insulin users: AID can now be considered for people using basal insulin only who are not meeting their personalized blood sugar targets.
In short, if you’re on insulin and considering a pump, the 2026 guidelines strongly recommend starting with an AID system instead of a traditional pump. Concerns about low blood sugar are a common reason people hesitate. AID systems are specifically designed to reduce that risk.
How to Choose the Right Insulin Pump for Your Lifestyle
There’s no one-size-fits-all pump — the best pick depends on your priorities, lifestyle, and health needs. Here’s a quick guide to help narrow it down.
| If you prioritize… | Consider… |
|---|---|
| Smallest size | Tandem Mobi or twiist |
| Tubeless design | Omnipod 5 (with AID) or Omnipod Dash (manual) |
| Best water resistance | Omnipod 5/Dash or Tandem Mobi (both IP28) |
| No carb counting required | iLet Bionic Pancreas (meal size only) or twiist with Diabeloop DBLG2 (2026) |
| Large reservoir (300 units) | Tandem t:slim X2, Medtronic 780G, twiist, Dana IIS |
| Apple Watch control (iPhone required) | twiist |
| Type 2 diabetes with AID | Omnipod 5 or Medtronic 780G (both FDA-cleared for T2D adults) |
| FreeStyle Libre CGM | Tandem t:slim X2, iLet, twiist, Omnipod 5 (verify Libre 3 Plus availability) |
| Finest basal increments | Medtronic 780G (0.025 units/hr minimum) |
Helpful Resources & Research
- Understanding diabetes complications and how better blood sugar control helps prevent them — DiabetesNet.com
- ADA Standards of Care in Diabetes — 2026, Section 7 (Diabetes Technology)
- Integrated Diabetes Services 2026 In-Depth Pump Comparison Chart
- Tidepool Loop algorithm information
- Beta Bionics iLet information
- Omnipod 5 current compatibility resources
- Tandem Diabetes pump specifications
- twiist AID system
What is an insulin pump and how does it work?
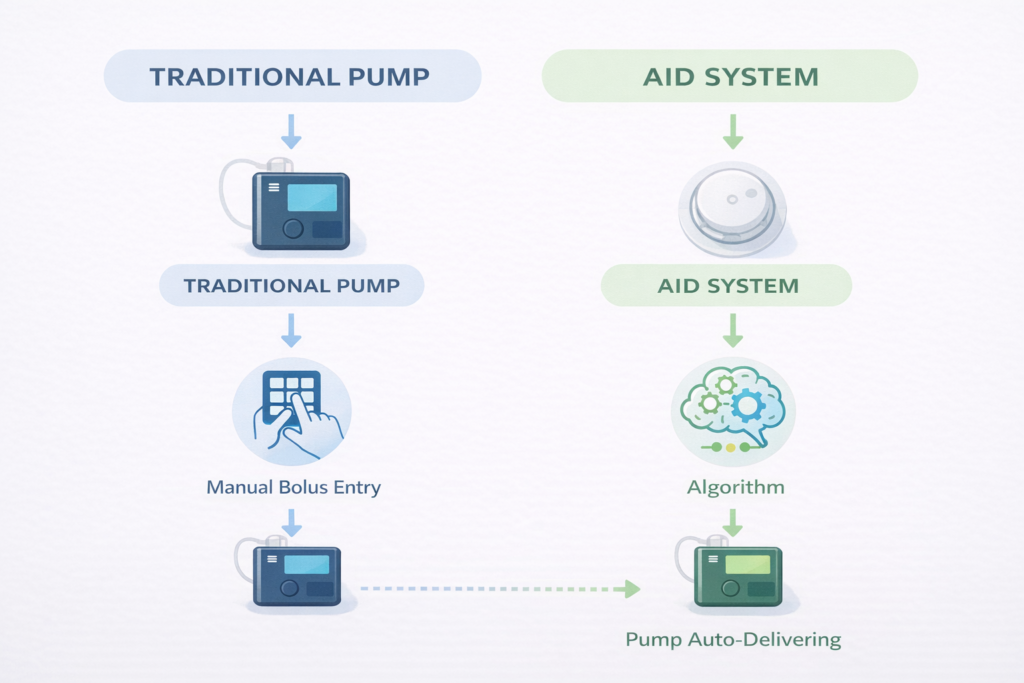
An insulin pump is a small, wearable device that continuously delivers rapid-acting insulin throughout the day, replacing the need for multiple daily injections. It provides a small background dose (basal rate) around the clock and additional doses (boluses) at mealtimes or to correct high blood sugar. Modern pumps connect to a continuous glucose monitor (CGM) and use a computer algorithm to automatically adjust insulin delivery — these are called automated insulin delivery (AID) systems.
What distinguishes an AID system from a traditional insulin pump?
A traditional pump delivers insulin based on fixed settings you program. An AID system — also called a closed-loop or hybrid closed-loop system — connects your pump to a CGM and uses an algorithm to automatically adjust your insulin every few minutes based on real-time blood sugar readings. AID systems greatly reduce the daily burden of diabetes management and are now the standard of care recommended by the ADA for eligible people (2026).
Which insulin pump offers the least daily management?
The iLet Bionic Pancreas requires the least daily input — you only indicate the size of each meal (small, medium, or large) instead of counting carbohydrates. The twiist pump also automates most adjustments, and in 2026, its optional Diabeloop DBLG2 algorithm only needs your weight and total daily insulin — no meal entry required. In most other AID systems, you still need to announce meals by entering carbohydrate counts.
Which insulin pumps are compatible with FreeStyle Libre CGMs?
As of 2026, the Tandem t:slim X2, iLet Bionic Pancreas, and twiist are compatible with the FreeStyle Libre 3 Plus in the U.S. Omnipod 5 currently works with the Libre 2 Plus, and FreeStyle Libre 3 Plus rollout is in progress — check current Omnipod resources to confirm availability before prescribing. The Tandem Mobi does not currently support FreeStyle Libre sensors. The Medtronic 780G is only compatible with Medtronic’s own CGM sensors.
What blood sugar targets can insulin pumps be set to?

Most AID systems have a target range of approximately 100–180 mg/dL (5.6–10.0 mmol/L). The twiist offers the widest range — from as low as 87 mg/dL (4.8 mmol/L) to 180 mg/dL (10.0 mmol/L). The Medtronic 780G aims for around 100 mg/dL (5.6 mmol/L) during active AID. The Omnipod 5’s upcoming 2026 algorithm update is expected to lower its available target to 100 mg/dL (5.6 mmol/L). Always set targets with guidance from your care team.
Are insulin pumps covered by insurance or Medicare?
Most major insurance plans and Medicare Part B cover insulin pumps for people with type 1 diabetes who meet clinical criteria. Coverage for type 2 diabetes has expanded following FDA clearances for Omnipod 5 and Medtronic 780G in people with T2D on insulin. The twiist is designed to be dispensed through retail pharmacies, which may simplify access and lower upfront costs. Always verify your specific plan’s coverage before choosing a pump.
What is the newest insulin pump on the market in 2026?
The twiist AID System from Sequel Med Tech launched in the U.S. on July 7, 2025, making it the newest commercially available AID pump. It uses sound-wave technology to measure every microdose of insulin, sets the lowest available blood sugar target (87 mg/dL / 4.8 mmol/L), and is the first FDA-cleared AID system operable from an Apple Watch (with an iPhone as the primary device).
How often should you change an insulin pump infusion set or pod?
Traditional tubed pump infusion sets are usually replaced every 2–3 days. Tandem received FDA clearance in late 2025 for extended-wear use of its SteadiSet infusion set, with a broader commercial rollout expected afterward. Omnipod pods are changed every 3 days. The Medtronic Extended infusion set can last up to 7 days on compatible pumps. The twiist’s disposable insulin cassette is swapped out every 3 days, while the reusable pump body lasts up to 3 years.
Which insulin pump has the best waterproofing?
The Omnipod 5, Omnipod Dash, and Tandem Mobi all have an IP28 rating. Omnipod pods are tested to withstand submersion up to 25 feet for 60 minutes, making them the best choice for swimmers. The Tandem Mobi is rated for 8 feet for 2 hours. The Medtronic 780G has a much lower IP22 rating, protecting only against dripping water — it is not suitable for swimming or water sports.
Can people with type 2 diabetes use an insulin pump?
Yes — both the Omnipod 5 and Medtronic 780G are now FDA-cleared for adults with type 2 diabetes who use insulin. The Omnipod Dash is also approved for anyone needing insulin. The ADA’s 2026 Standards of Care support AID use in type 2 diabetes for people on multiple daily injections who are not achieving their blood sugar targets, and research is ongoing for fully automated systems designed specifically for type 2 management.
Last Updated on April 7, 2026