Update: FreeStyle Navigator® Continuous Glucose Monitoring System U.S. Customers
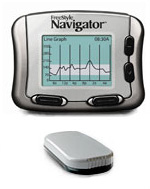
The Food and Drug Administration (FDA) has approved the FreeStyle Navigator® Continuous Glucose Monitoring System in the United States for people with diabetes. Designed to discretely and continuously measure glucose levels through a sensor in the back of the upper arm or abdomen, this system provides minute-by-minute information about which way and how quickly blood sugar levels are changing. This information can lead to adjustments that can result in tighter glucose ranges. As with all current continuous monitors, a traditional fingerstick glucose test should be performed before making any treatment decisions.
- Step 1: Complete and sign an Authorization to Release Patient Information.
- Step 2: Complete and sign the Patient Information Questionnaire.
- Step 3: Obtain a completed Statement of Medical Necessity from your physician.
- Step 4: Submit your information to the ADC Resource Center.
For more information about each of these steps, please visit Abbott’s purchase page.
- 1 Receiver
- User’s Guide
- Getting Started Guide
- Welcome Card
- 6 Overbandages
- 6 IV Wipes
- 6 Alcohol Prep Pads
- 30 Sterile Lancets
- Quick Reference Card
- Freestyle Strip Insert
- Freestyle Control Solution Insert
- Warranty Registration Card
- Transmitter and Accessory Kit – which inludes 1 belt clip, 1 transmitter, 1 lancing device with clear and grey cap, 2 AAA alkaline batteries for the receiver, 1 vial of Freestyle Control Solution, 1 vial of 50 Freestyle Strips, 1 Silver Oxide 357 HC Battery for the Transmitte
Some Issues:
- Batteries fall out of the receiver. Taping over or putting a rubber band around the cover can help avoid this.
- Sensor is lost if batteries fall out of the receiver.
- When BG goes high or low, alerts sound every 10 min while the BG remains above or below the selected target.
- Poor adhesive
- Display does not light up continuously. Requires you to push 2 buttons to light it.
Last Updated on October 15, 2019
