The duration of insulin action (DIA), also called active insulin time, is one of the most important settings in your insulin pump, yet it is also among the most misunderstood. DIA tells your pump how long a bolus of insulin continues to lower your blood sugar. When your pump knows this, it can accurately calculate how much insulin is still working in your body (called insulin on board, or IOB), so it does not recommend too much insulin the next time you bolus.
Most people set their DIA too short, often 2 to 3 hours, because they believe their rapid-acting insulin works quickly. For many users of standard rapid-acting analogs, a longer setting, usually around 4.5 to 5 hours and sometimes longer depending on the system and insulin, better aligns with real-world insulin activity than 2 to 3 hours. Setting DIA too short can be a significant and often overlooked cause of insulin stacking and over-correction. This article explains what DIA truly is, how to choose a setting that fits your situation, how automated insulin delivery (AID) systems manage DIA differently from traditional pumps, and what to do if you think your DIA has been set too short.
Jump to: Frequently Asked Questions about Duration of Insulin Action
Quick Reference: DIA Starting Ranges by Insulin Type
Use the table below as a starting point and adjust it with your diabetes care team. The appropriate setting depends on your insulin, your pump or AID system, and your individual glucose patterns.
| Insulin Type | Starting DIA Range | Notes |
|---|---|---|
| Rapid-acting (Humalog, Novolog/NovoRapid, Apidra) | ~4.5–5 hrs | Supported by published pharmacodynamic studies and most commercial algorithm defaults |
| Ultra-rapid (Lyumjev, Fiasp) | ~4–5 hrs | Faster onset and earlier peak; meaningful insulin tail still extends well past 3 hrs |
| Regular (U-100) | ~5.5–6.5 hrs | Slower overall profile; rarely used in current pump therapy |
What is the duration of insulin action (DIA)?
DIA is the total time from when you deliver a bolus until that bolus no longer lowers your blood sugar. Think of it as the lifespan of a single dose of insulin in your body. The DIA timer starts the moment you give a bolus and ends only when that insulin stops affecting your glucose.
Your pump uses your DIA setting to calculate insulin on board (IOB), the amount of bolus insulin still active from previous doses. When you go to bolus again, the pump subtracts your current IOB from the recommended dose. This helps prevent insulin stacking: giving a new bolus on top of insulin that is still working.
In a study involving 396 insulin pump users, 64% of over 132,000 bolus doses were administered within 4.5 hours of a previous bolus.1 This indicates that most boluses overlap with residual insulin. An accurate DIA prevents your pump from delivering too much insulin on top of what is already active.
How long does rapid-acting insulin typically last in your body?
Here is where much confusion begins. Package inserts for rapid-acting insulins like Humalog, Novolog, and Apidra list an insulin action time (IAT) of “3 to 5 hours.” Many pump users, and even some clinicians, use this number directly when setting DIA, but these two values answer different questions.
Package-insert IAT and pump DIA are different things. Package-insert IAT is measured under standardized pharmacologic conditions, with specific dose sizes and glucose-clamp methods designed to compare insulin products. These values are scientifically accurate for what they measure, but they don’t answer the same question a pump bolus calculator needs to. The calculator must know: how long will this bolus lower this patient’s blood sugar in real-world conditions? Package-insert studies usually don’t include the early minutes before glucose-lowering effects start, use dose sizes and measurement endpoints that can make the effective duration seem shorter, and are not meant to set pump parameters.
Published pharmacodynamic studies of subcutaneously injected rapid-acting analogs, including Humalog, Novolog, and Apidra, have shown insulin activity with a significant tail extending well past 3 hours, with many studies reporting glucose-lowering effects out to approximately 5 hours or longer, depending on dose, method, and individual.2,3,4 These findings have shaped the 4- to 5-hour defaults used in several commercial pump algorithms, including Tandem Control-IQ’s fixed 5-hour active insulin time. Even ultra-rapid formulations (Lyumjev, Fiasp) show meaningful activity beyond 3 hours, with pharmacodynamic profiles that support setting DIA to at least 4 hours.5
Practical example: A 7-unit dose of Lyumjev measured an insulin action time (IAT) of 4.6 hours in published clinical testing,5 and that figure does not include about 17 minutes between injection and the start of the glucose clamp measurement. The actual duration of the glucose-lowering effect is even longer.
Why does my insulin seem to work more quickly than that?
This is a common experience, and there are several reasons why insulin might appear to act faster than it actually does.
- Insulin stacking from a previous bolus: If your blood sugar drops 30 minutes after a bolus, the likely cause is residual insulin from an earlier dose, not the bolus you just administered.
- Overestimating carbs or eating low-glycemic foods: A bolus that is too large for the meal will cause a sharp drop, making the insulin seem to act too quickly.
- High total daily dose: Taking too much insulin overall can cause frequent lows, making insulin seem to act quickly.
- Physical activity: Exercise alone lowers blood sugar. A post-bolus workout can make insulin seem to work faster when activity is doing most of the work.
Parents of children with diabetes are especially vulnerable to these misconceptions. Because children are more sensitive to insulin, a half-unit too much can cause a dramatic drop that appears similar to fast-acting insulin, when the real issue is dose sensitivity. In a glucose clamp study of 11 young children, researcher Dr. Bruce Buckingham at Stanford found that instead of working quickly, their insulin acted more slowly than typical published data for adults6, directly the opposite of what many parents believe.
Is the duration of insulin action different in children compared to adults?
Available pediatric data do not support routinely using shorter DIA settings in children than in adults. Several small studies have found broadly similar pharmacodynamic profiles across age groups, and at least one study found children’s active insulin time to be somewhat longer than in adults, not shorter. However, the evidence base is limited in size and methodological consistency, and these studies should not be read as proof that DIA is identical across all children and adults.
- A study of 18 children aged 6 to 18 years concluded that Novolog (aspart) pharmacodynamics in children and adolescents were similar to those in adults with Type 1 diabetes.7
- A study of 9 children aged 6 to 12 years found no significant difference in insulin action time compared to adults.8
- A Stanford study of 8 young children found their DIA was slightly longer than adult values, not shorter.6
- A study of Apidra (glulisine) in 20 children aged 7 to 16 found no difference in glucose-lowering duration compared to adults.9
The practical lesson: if a child’s insulin seems to work very quickly, factors like dose sensitivity, activity, and leftover insulin from a previous bolus are more likely explanations than a truly faster insulin profile.
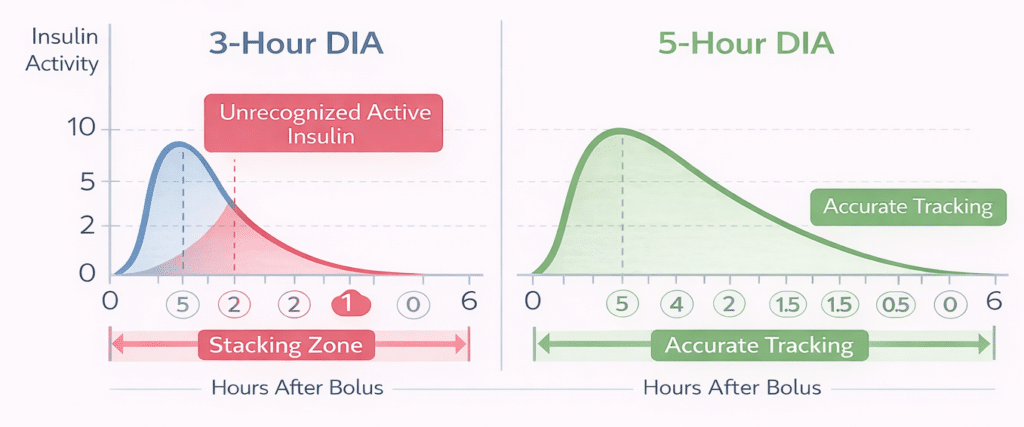
Why is a short DIA setting problematic?
Setting your DIA too short, for example 2 or 3 hours, can be an important and often overlooked cause of insulin stacking, over-correction, and unexplained lows. The issue is difficult to detect because the root cause is hidden within your pump settings, and the symptoms may seem to have other explanations.
How a short DIA causes insulin stacking
When your DIA is set to 3 hours, your pump assumes all insulin from a bolus has been used up after that time. So, if your blood sugar is high at the 3-hour mark, the pump suggests a full correction dose, ignoring the insulin still active from the earlier bolus.
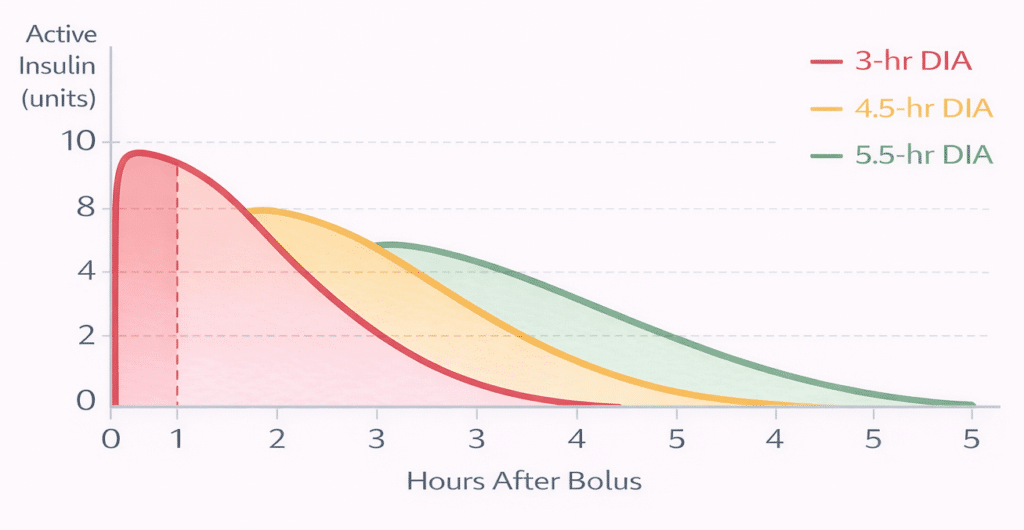
Example: You give a breakfast bolus at 7:00 AM. At 10:00 AM, your blood sugar remains high. With a 3-hour DIA, your pump reports no active insulin and recommends a full correction dose. But with a 5-hour DIA, your pump recognizes that a significant part of the breakfast bolus is still active and suggests a smaller, or no, correction. The table below shows calculated IOB at each hour for a 10-unit bolus under three different DIA settings.
| DIA Setting | 0 hrs | 1 hr | 2 hrs | 3 hrs | 4 hrs | 5 hrs |
|---|---|---|---|---|---|---|
| 3-hr DIA | 10 u | 6.7 u | 3.3 u | 0 u | 0 u | 0 u |
| 4.5-hr DIA | 10 u | 7.8 u | 5.6 u | 3.3 u | 1.1 u | 0 u |
| 5.5-hr DIA | 10 u | 8.2 u | 6.4 u | 4.5 u | 2.7 u | 0.9 u |
With a 3-hour DIA after a 10-unit bolus, the pump shows no active insulin at the 3-hour mark. A 5.5-hour DIA setting would suggest about 4.5 units remaining active at that same time. The difference between these two estimates directly affects all subsequent bolus recommendations.
The domino effect of a short DIA
Short DIA settings often cause a frustrating cycle that is difficult to trace back to the original setting.
- Step 1: Short DIA hides IOB, and the pump recommends too much insulin
- Step 2: Excess bolus insulin causes low blood sugar
- Step 3: Lows are blamed on basal rates being too high
- Step 4: Basal rates are lowered to compensate
- Step 5: Lower basal causes high blood sugar between meals
- Step 6: High readings are managed with larger correction boluses
Unless someone identifies the short DIA as the starting point, this cycle continues, and other settings keep being adjusted in the wrong direction. If you correct your DIA, you might need to revisit basal rates and carb ratios that were previously changed to compensate.
How do various insulin pumps determine insulin on board?
Not all pumps process DIA and IOB the same way, which has caused confusion for people switching pumps or working with clinicians unfamiliar with a specific device.
Linear versus curved decline: Some pumps, such as Accu-Chek and Omnipod in manual mode, estimate insulin activity as a straight-line decrease, for example, 20% or 25% per hour. Others, including Medtronic and Tandem, use a curved decline that more accurately reflects the pharmacodynamic shape of rapid-acting insulin. Curved models generally provide more precise IOB calculations over the entire duration.
Note — legacy system comparison (historical context only): Older pump models from different manufacturers used different baseline reference points for their DIA scales, leading to confusion when clinicians tried to transfer settings between devices. For example, a 3.5-hour setting on the discontinued Animas 1200 was roughly equivalent to 8 hours on a Medtronic Paradigm and 5.5 hours on a Cozmo pump.10 These specific cross-pump equivalencies apply only to those discontinued platforms and should not be applied to current pumps or AID systems. They are included here because they help explain why so many pump users ended up with DIA settings far shorter than they should have been, and why that pattern persists today.
For help understanding how your current pump’s IOB calculation works and how it integrates with continuous glucose monitoring, see our guides on insulin pumps and CGMs.
What DIA setting should I select for my insulin pump?
For most people using standard rapid-acting insulin (Humalog, Novolog, Apidra) in a traditional pump, a starting DIA of about 4.5 to 5 hours generally aligns with available pharmacodynamic data and the defaults used by many commercial algorithms. If you use an ultra-rapid insulin (Lyumjev or Fiasp), a starting point of 4 to 5 hours is reasonable; these insulins peak earlier but still have a significant tail past 3 hours. The best setting for you depends on your specific insulin, pump, and glucose patterns.
Real-world pump download data indicate that short DIA settings are common. Analyzing over 1,000 U.S. pump downloads in 2007 revealed an average DIA setting of 3.1 hours,1 and a 2014 review of more than 1,000 U.S. and European pump datasets found an average of 3.4 hours.10 Both figures are below the ranges supported by pharmacodynamic literature, implying that many pump users operate with settings that lead to unrecognized insulin stacking.
Important: Do not shorten your DIA to get larger bolus recommendations. If your bolus calculator cannot lower high blood sugar to the target when IOB is present, adjust your basal rates or carb ratios instead. Using DIA as a workaround hides the true problem and makes it harder to correctly calibrate the rest of your settings.
What if I correct my DIA: what else should I check?
If you increase your DIA from 2 to 3 hours toward 4.5 to 5 hours, your pump will begin recognizing more active insulin and recommend smaller corrective doses. You might initially see higher blood sugar readings, especially in the afternoon and evening, if your basal rates or carb ratios were previously set higher to compensate for the short DIA. Work with your diabetes care team to reassess:
- Basal rates (especially afternoon and evening)
- Insulin-to-carb ratios
- Correction factor (insulin sensitivity factor)
Does DIA vary among individuals?
Yes, but not as much as most pump settings permit. One well-cited study of Novolog found about a 25% variation in insulin action time among different individuals.2 That sounds significant, but let’s put it into perspective:
If the average time to peak activity is about 2 hours, a 25% variation means it could peak anywhere between roughly 1 hour 45 minutes and 2 hours 15 minutes for most people. Since the total average duration is 5 to 6 hours, the variation among individuals would be roughly within that same proportional range, not the 2 to 8 hours allowed by current pump settings.
Today’s pumps permit DIA settings from 2 to 8 hours, a range much broader than the biological variation in the population. This extensive allowable range is one reason why short settings are so common and so tempting.
How do automated insulin delivery (AID) systems manage the duration of insulin action?
Automated insulin delivery (AID) systems combine an insulin pump with a continuous glucose monitor (CGM) and a control algorithm that adjusts insulin every 5 minutes based on glucose readings. According to the 2026 ADA Standards of Care, AID is now the preferred method of insulin delivery for people with Type 1 diabetes and is recommended for certain adults with Type 2 diabetes on insulin who are not meeting their goals.
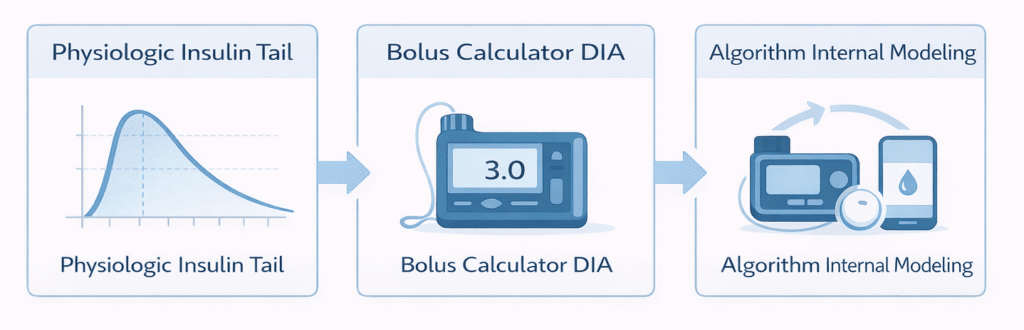
This is where DIA becomes much more complex. Not all AID systems use DIA in the same way, and in some cases, the user-entered DIA and the algorithm’s own internal insulin monitoring are completely separate. Before examining individual systems, it helps to understand three distinct concepts that these systems handle differently.
Three Concepts Every AID User Should Understand
| Concept | What it means for DIA |
|---|---|
| 1. Physiologic insulin tail | How long a real-world bolus actually lowers blood sugar, generally extending well past 3 hrs for rapid-acting analogs, often to around 5 hrs in many pharmacodynamic studies. |
| 2. Bolus-calculator DIA/AIT | The number you enter in your pump or AID system that tells the bolus calculator how much IOB remains from a user-given bolus. Should reflect the physiologic tail in traditional pumps and most open-source systems. |
| 3. Algorithm-internal insulin modeling | How the AID algorithm itself tracks or estimates insulin on board for automated delivery decisions. This is separate from the user-entered DIA; it varies by system and is not always visible or configurable. |
The confusion that occurs when switching between AID systems, or comparing them, almost always comes from mixing up these three concepts. For example, the Medtronic 780G recommends a short AIT setting of 2 to 3 hours, but that suggestion relates to Concept 2 (how the bolus calculator scales autocorrections), not Concept 1 (the physiologic insulin tail). The physiologic tail remains unchanged. And Concept 3 (the algorithm’s internal modeling) works independently of both.
Tandem Control-IQ (t:slim X2 and Mobi)
When Control-IQ is active, it sets the bolus-calculator’s active insulin time to 5 hours, regardless of your pump profile settings. This choice was based on clinical trial data showing it minimizes hypoglycemia while keeping a strong time in range.11 Tandem’s algorithm also uses this value as part of its automated delivery modeling (Concept 3), ensuring Control-IQ’s DIA management is consistent in both bolus calculation and automated adjustments.
The DIA setting you programmed into your pump profile only applies when you turn Control-IQ off and switch back to manual mode. When the algorithm is active, your programmed DIA remains inactive.
Medtronic MiniMed 780G (SmartGuard)
The 780G calls its setting Active Insulin Time (AIT), but Medtronic explicitly states that it is not intended to reflect physiological insulin metabolism.12 In the 780G, AIT is a Concept 2 parameter: it controls how large the automatic correction boluses are, not how long the physiologic insulin tail extends.
Medtronic recommends an initial AIT of2 to 3 hours. A shorter AIT causes SmartGuard to deliver larger, more frequent autocorrections. Research in 235 real-world 780G users found that a 2-hour AIT significantly increased autocorrection delivery and improved time in range compared to longer settings.13 SmartGuard uses its own internal algorithm, tracking current glucose, trend, and rate of change, to manage stacking risk (Concept 3). It does not rely on a traditional IOB decay curve like a conventional pump.
Critical takeaway: The 780G’s short AIT recommendation is specific to SmartGuard’s algorithm. It does not imply that short DIA settings are suitable for traditional pumps or other AID systems, where a too-short setting can lead to insulin stacking.
Omnipod 5 (SmartAdjust)
Omnipod 5 uses a user-adjustable DIA, usually set between 3 and 4 hours, but this is a Concept 2 parameter only: it governs the IOB calculation for boluses the user delivers manually. The system’s automated insulin delivery (Concept 3) runs on a total daily insulin (TDI) model, not the user-entered DIA. Every 5 minutes, SmartAdjust looks 60 minutes ahead and adjusts automated delivery based on predicted glucose and daily insulin history.
For most users, the practical focus should be on accurate target glucose and TDI settings as the main drivers of automated performance, while keeping the bolus-calculator DIA at a clinically reasonable value for manual doses.
iLet Bionic Pancreas (Beta Bionics)
The iLet adopts the most hands-off approach among commercial AID systems: it requires no user-entered DIA at all. Setup only needs the user’s weight. The system then uses fully autonomous, self-learning algorithms to determine 100% of all insulin doses, including basal, correction, and meal-related doses, without any input of DIA, carb ratios, correction factors, or basal rates.
The iLet’s algorithms continuously adapt with each pod change by using delivery history and CGM data to update their internal models. All three concepts above are managed internally by the algorithm. A 2025 clinical trial published in Clinical Diabetes confirmed the effectiveness of the iLet when initiated by primary care providers, including via telehealth, with 97% of participants achieving an average blood sugar below 183 mg/dL (10.2 mmol/L).14
Open-source AID systems (Loop, AndroidAPS, Trio, twiist)
Open-source AID systems, including Loop, AndroidAPS, Trio, and the FDA-cleared twiist (powered by the Tidepool Loop algorithm), all require a user-entered DIA as part of their core insulin modeling. In these systems, the user-entered DIA is a full Concept 1/2/3 value: it drives both the bolus calculator IOB and the algorithm’s internal predictions. The 2026 ADA Standards of Care now explicitly recognize open-source systems as a legitimate option in diabetes care.
Unlike commercial systems that replace or ignore the user-entered DIA, open-source systems use it directly in their glucose prediction models. The right value varies depending on the specific algorithm (Loop, oref0/oref1 used in AndroidAPS and Trio, etc.) and the insulin type. For standard rapid-acting analogs, roughly 5 hours is a common starting point in community practice and available documentation, but this should be seen as a starting estimate, not a universal rule, and confirmed with your care team or the system’s documentation.
AID System DIA Comparison Table
Use this table as a quick reference when starting a new system or advising someone who is switching. Note that the correct DIA entry for one system might not be right for another.
| AID System | DIA/AIT Setting | User Adjustable? | Key Consideration |
|---|---|---|---|
| Tandem Control-IQ (t:slim X2 / Mobi) | Fixed at 5 hrs | No (automated mode) | Locked to a physiologically grounded value; your programmed DIA applies only in manual mode. |
| Medtronic MiniMed 780G (SmartGuard) | 2–3 hrs recommended | Yes (2–8 hrs range) | AIT here is NOT physiological DIA; it controls autocorrection magnitude. Medtronic explicitly states this. |
| Omnipod 5 (SmartAdjust) | 3–4 hrs typical | Yes | User-entered DIA governs IOB from manually delivered boluses only; automated delivery uses its own internal accrual model. |
| iLet Bionic Pancreas (Beta Bionics) | Not applicable | No setting exists | No DIA required. All dosing is determined autonomously from user weight and delivery history. |
| Open-source systems (Loop, AndroidAPS, Trio, twiist) | ~5 hrs common start | Yes (full control) | DIA drives IOB modeling; optimal value depends on the specific algorithm and insulin. Undersetting causes stacking. |
DIA tips for switching to another AID system
Switching AID systems is becoming more common as new options enter the market. Your DIA setting from one system shouldn’t automatically transfer to the next, as each system uses that parameter differently.
- Traditional pump to Control-IQ: DIA automatically locks at 5 hours when the algorithm is active. No action needed.
- Control-IQ to 780G: Do not import your 5-hour DIA as AIT. Start at Medtronic’s recommended 2 to 3 hours and adjust based on glucose outcomes.
- Any system to Omnipod 5: DIA manages IOB solely based on your manual boluses. Focus on setting an accurate target glucose and establishing a solid total daily insulin baseline for the automated algorithm.
- Any system to iLet: No DIA entry needed. The system creates its own delivery model based on your weight and adapts over time.
- Starting Loop, AndroidAPS, Trio, or twiist: DIA drives all insulin modeling in these systems. See the open-source section above for starting range guidance, and confirm your setting with your care team and the system’s documentation.
When unsure, contact the manufacturer’s clinical support or your diabetes care team before adjusting key settings. AID systems are complex, and small changes to core parameters can significantly impact insulin delivery.
Helpful Resources & Research
- ADA Standards of Care 2026
- FDA Insulin Pump Guidance
- Time-Action Profiles of Insulin Preparations
- Pumping Insulin, 7th Edition
- Clinical Implementation of the Omnipod 5 Automated Insulin Delivery System
- Assessing the iLet Bionic Pancreas Deployed in Primary Care and via Telehealth
- ClinicalTrials.gov
Frequently Asked Questions: Duration of Insulin Action
What is the meaning of insulin action duration (DIA)?
DIA is the total duration that a bolus of rapid-acting insulin continues to lower your blood sugar. It begins when you inject or deliver the bolus and ends when that dose is no longer active. Your insulin pump uses this number to estimate how much insulin is still working from previous doses (called insulin on board, or IOB), so it doesn’t recommend too much insulin for your next bolus.
What DIA setting is recommended for most pump users to start with?
For most people using standard rapid-acting insulin (Humalog, Novolog, or Apidra) in a traditional pump, a starting range of about 4.5 to 5 hours generally aligns with available pharmacodynamic data and the defaults used by many commercial algorithms. If you use Lyumjev or Fiasp, starting around 4 to 5 hours is reasonable. The appropriate setting depends on your insulin, your system, and your individual glucose patterns — always confirm with your diabetes care team.
Why does my insulin seem to work in only 2 to 3 hours?
Several factors can give the impression that insulin acts faster than it actually does. Residual insulin from a previous bolus is a common reason for a quick drop after a new bolus. You might also be overestimating carbs, taking too much insulin overall, or exercising after a bolus. In each case, it’s not that the insulin is inherently fast — something else is causing your blood sugar to drop more quickly than expected.
What is insulin stacking and why does it pose a problem?
Insulin stacking occurs when you administer a new bolus before the previous one has finished working. If your DIA is set too short, your pump underestimates the amount of insulin still active and recommends doses that are too large. The result is blood sugar that drops lower than intended — sometimes causing a low that seems to come out of nowhere. An accurate DIA provides your pump with a realistic view of active insulin, leading to more appropriate recommendations.
Why can’t I just rely on the number listed on the insulin package insert?
Package-insert action times are measured under standardized pharmacologic study conditions designed to compare insulin products with each other — they answer a different question than a pump bolus calculator is trying to answer. The package-insert value is scientifically valid for its purpose, but it is not interchangeable with a pump DIA setting. Copying package-insert numbers directly into your pump DIA is likely to produce a setting that’s too short for accurate real-world bolus IOB tracking.
Is the duration of insulin action different for children compared to adults?
Available data do not support routinely setting a shorter DIA in children than in adults. Several small studies found broadly similar pharmacodynamic profiles across age groups, and one study found children’s insulin worked slightly slower than adults, not faster. The evidence base is limited, but the practical guidance is consistent: if a child’s blood sugar drops quickly after a bolus, factors like dose sensitivity, physical activity, or residual insulin from a previous bolus are more likely causes than a genuinely shorter insulin duration.
What are the effects of shortening my DIA to get larger bolus recommendations?
Shortening DIA to push for larger recommended doses is a common but problematic workaround. A shorter DIA causes your pump to think less insulin is still active, leading it to recommend bigger boluses. This hides the true cause of high blood sugar and raises the risk of insulin stacking and lows. If your pump’s suggestions don’t bring blood sugar to target, work with your care team to adjust basal rates or insulin-to-carb ratios instead.
Do all insulin pumps calculate insulin on board the same way?
Pumps vary in the DIA scale they use and how they model IOB. Some use a straight-line decline; others use a curve that better matches the pharmacodynamic profile of rapid-acting insulin. Older pump models used entirely different reference points, so a “3.5-hour” setting on one discontinued platform was equivalent to an “8-hour” setting on another. See the AID System DIA Comparison Table for how current systems compare.
How does DIA work differently in automated insulin delivery (AID) systems?
Each AID system manages DIA differently. Tandem Control-IQ sets active insulin time at 5 hours when the algorithm runs. The Medtronic 780G uses a setting called Active Insulin Time (AIT), intentionally set short (2 to 3 hours) to manage autocorrection levels — Medtronic explicitly states this does not reflect the actual physiological insulin duration. Omnipod 5 uses the user-entered DIA only for IOB from manually delivered boluses; its automated delivery relies on its own internal model. The iLet Bionic Pancreas has no user-entered DIA at all. Open-source systems use the user-entered DIA directly in their insulin modeling, with around 5 hours being a common starting point for rapid-acting analogs.
What should I check after updating my DIA setting?
When you increase your DIA to a longer, more evidence-supported range, your pump will identify more active insulin and suggest smaller correction doses. You might initially see higher blood sugar levels — especially in the afternoon and evening — if your basal rates or carb ratios were previously set higher to account for the shorter DIA. Have your diabetes care team review your basal rates, insulin-to-carb ratios, and correction factor after updating your DIA.
References
- Walsh J, Wroblewski D, Bailey TS. Disparate Bolus Recommendations in Insulin Pump Therapy. Poster, AACE Meeting, 2007.
- Heinemann L, Weyer C, Rauhaus M, Heinrichs S, Heise T. Variability of the metabolic effect of soluble insulin and the rapid-acting insulin analog insulin aspart. Diabetes Care. 1998;21(11):1910–1914.
- Heinemann L. Time-Action Profiles of Insulin Preparations. Kirchheim & C. GmbH, Mainz, Germany, 2004.
- Rave K, Nosek L, de la Pena A, et al. Dose response of inhaled dry-powder insulin and dose equivalence to subcutaneous insulin lispro. Diabetes Care. 2005;28(10):2400–2405.
- Becker RHA, et al. [Lyumjev IAT reference; see also Lilly prescribing information for Lyumjev, 2020.] Diabetes Obes Metab. 2015;17:261–267.
- Buckingham BA, Block J, Wilson D, Rebrin K, Steil G. Novolog Pharmacodynamics in Toddlers. Diabetes. 2005;54(Suppl 1):Abstract 1889-P.
- NovoLog Physician Insert. Approved October 21, 2005. [Pediatric pharmacodynamic data, ages 6–18.]
- Mortensen HB, Lindholm A, Olsen BS, Hylleberg B. Rapid appearance and onset of action of insulin aspart in paediatric subjects with type 1 diabetes. Eur J Pediatr. 2000;159(7):483–488.
- Becker R, Frick A, Heinemann L, Nosek L, Rave K. Dose-response relation of insulin glulisine in subjects with type 1 diabetes. Diabetes. 2005;54(Suppl 1):A332 (Abstract 1367-P).
- Walsh J, Roberts R. Pumping Insulin, 5th ed. Torrey Pines Press, 2013. [Cross-pump DIA equivalency tables, pp. 93–112; 2014 Diasend DIA average data, private communication.]
- Breton MD, Kanapka LG, Beck RW, et al. A Randomized Trial of Closed-Loop Control in Children with Type 1 Diabetes. N Engl J Med. 2020;383(9):836–845.
- Medtronic. Adjusting Active Insulin Time — MiniMed 780G System Support. medtronicdiabetes.com (accessed 2025).
- Lopez-Masso D, et al. Active insulin time setting in MiniMed 780G: Impact on glucose control and patient perception. Endocrinol Diabetes Nutr. 2025.
- Oser SM, Putman MS, Russell SJ, et al. Assessing the iLet Bionic Pancreas Deployed in Primary Care and via Telehealth: A Randomized Clinical Trial. Clin Diabetes. 2025;43(3):388–398.
Last Updated on March 25, 2026