SpectRx, Inc. is developing a non-invasive test to screen for diabetes by measuring the intensity of fluorescence and scattering in the lens of the eye. Eye measurements are performed through an undilated pupil with the assistance of a pupil tracking system.
The SpectRx diabetes screening test is designed to be painless, provide easy access, equivalent accuracy, take approximately 20 seconds and will not require that the person be fasting. It is designed as a desktop system for use in medical settings to provide high quality results at costs below conventional screening methods. The technology has been licensed to (Boehringer Mannheim Corporation BMC) which is now owned by Roche. SpectRx estimates there are 50 million undiagnosed people with diabetes worldwide.
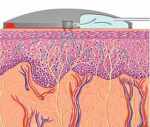
SpectRX is also developing the Altea MicroPor(TM) Laser which creates microscopic pores in the stratum corneum, the outermost layer of skin. This painless proceedure opens paths for interstitial fluid (ISF) to cross the outer skin barrier. Glucose can then by measured in the ISF that is collected in an external patch on the skin which contains a glucose sensor.
The laser device uses a low-cost, low-energy laser with a self-regulating feature to insure no damage occurs to viable tissues beyond the outer layer of dead skin cells. Being on the skins surface, the sensor aviods accuracy complications related to implanted sensors below the skin. Correlation between the ISF measured by this device and blood glucose measured with a commercial analyzer, over a three day period, was best on the first day at 0.95 gradually declining to 0.91 on day three. This device could also be rapidly adapted to Kumetrix's microneedle technology.
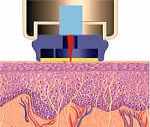
Research by SpectRx shows a direct correlation between glucose in the ISF and in blood, and that glucose in pure ISF can be measured with existing, off-the-shelf glucose test strip technology. The SpectRx personal glucose monitoring technology has been licensed to Abbott Laboratories, the world's leading in-vitro diagnostic company. Abbott appears to be satisfied with progress as they continue to increase research and development funding in SpectRx.
Data on SpectRx's meter correlates closely with current meters. SpectRx reports a correlation coefficient of 0.90 in the 60-400 mg/dl glucose range. The clinical results were from 280 comparisons involving 20 different people with diabetes. SpectRx's device is currently calibrated from a standard meter, but the company is attempting to bypass this need for calibration entirely.
Last Updated on March 18, 2011